 
When they do, they become monatomic ions. We can use this observation to help us write the formula of an ionic compound. Thus, ionic compounds are electrically neutral overall, even though they contain positive and negative ions. As you can see in the figure below, there are no individual NaCl particles in the array instead, there is a continuous lattice of alternating sodium and chloride ions. Some elements, especially transition metals, can form ions of multiple charges. Ionic compounds exist as alternating positive and negative ions in regular, three-dimensional arrays called crystals. Chlorine makes ionic compounds in which the chloride ion always has a 1 charge. During the reaction to produce an ionic compound, the metal atom loses electrons and forms a positive ion. 
For example, sodium makes ionic compounds in which the sodium ion always has a 1+ charge. Ionic bonds form instead of covalent bonds when there is a large difference in electronegativity. In every ionic compound, the total number of positive charges of the cations equals the total number of negative charges of the anions. Most of the elements that make ionic compounds form an ion that has a characteristic charge. The strong attraction between positive and negative ions often produce crystalline solids that have high melting points. Individual atoms can gain or lose electrons. Ionic compounds form when positive and negative ions share electrons and form an ionic bond. 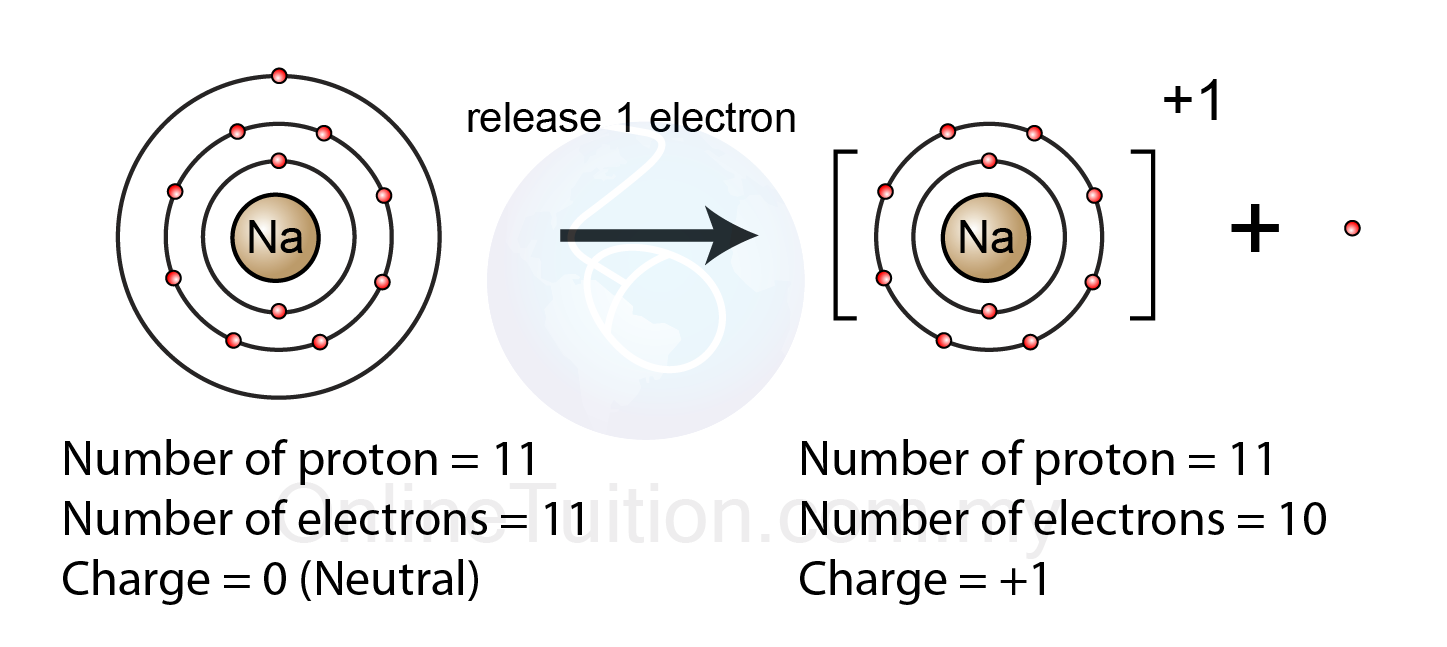
Compounds formed from positive and negative ions are called ionic compounds. In the chemical formula, the sum of the charges of the cations must always equal the sum of. (Remember from our conventions for writing formulas that we do not write a 1 subscript if there is only one atom of a particular element present.\): Naming the \(FeCl_2\) and \(FeCl_3\) Compounds in the Old/Common System. Remember that ions are formed only when electrons move from one atom to another a proton never moves from one atom to another. Ionic compounds always contain positive and. In ionic compounds, positive ions are surrounded by negative ions and vice versa. 
A cation is a positively charged ion and the anion is a negatively charged ion. Ions form when atoms lose or gain electrons to obtain a full outer shell: metal atoms lose electrons to form positively. , What mineral is composed of sodium chloride (NaCl), What best describes the general structure of an ionic compound and more. There are two types of ions: cation and anion. An ion is an atom or group of atoms with a positive or negative charge. A metal reacts with a nonmetal to form an ionic bond. When atoms gain or lose electrons, they usually gain or lose a characteristic number of electrons and so take on a characteristic overall charge. Individual atoms can gain or lose electrons. The formula Na 2Cl 2 also has balanced charges, but the convention is to use the lowest ratio of ions, which would be one of each. Study with Quizlet and memorize flashcards containing terms like The lattice energy of an ionic compound depends upon the. The electrostatic attraction between the positives and negatives brings the particles together and creates an ionic compound, such as sodium chloride. Compounds formed from positive and negative ions are called ionic compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
